|
Neutrons can be added or removed from an atom and the electrons around the atom really don't care that much. Neutrons don't change things greatly because they do not carry an electrical charge. Adding or removing protons changes the number and arrangement of electrons in the outer shell which changes how that atom reacts with other atoms. An atoms nucleus contains neutrons and protons. The number of electrons in an atom's outer shell determines the atom's chemical properties. This indicates that each chlorine atom contains 17 protons and must also have 17 electrons. Each proton in an atom's nucleus must be balanced with a negatively charged electron in one of the 'shells' outside the nucleus. Why does changing the number of protons in an atom change which element that atom is but changing the number of neutrons doesn't? Protons carry a positive charge. An atom of chlorine-35 contains 18 neutrons (17 protons 18 neutrons = 35 particles in the nucleus) while an atom of chlorine-37 contains 20 neutrons (17 protons 20 neutrons = 37 particles in the nucleus).Īdding or removing a neutron from an atom's nucleus creates isotopes of a particular element. Since all atoms of chlorine contain 17 protons, chlorine-35 and chlorine-37 differ in the number of neutrons each one has. The mass number is a tally of the number of protons and the number of neutrons in an atom's nucleus. The number after the name 'chlorine' is called the mass number. Adding or removing a proton from an atom's nucleus changes that atom's atomic number and creates a different element.Ĭhlorine-35 and chlorine-37 are both isotopes of the element chlorine. All atoms which contain 17 protons are called chlorine atoms. 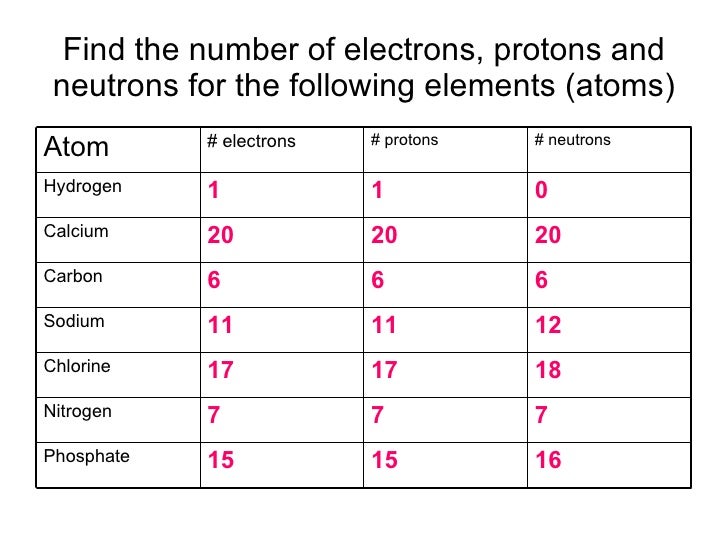
The number of protons an atom has, also known as the atom's atomic number, determines which element it is. All isotopes of an element behave the same way chemically for the most part.Why aren't Chlorine-35 and Chlorine-37 two different elements?Ĭhlorine-35 and chlorine-37 are not different elements because an atom of chlorine-35 and an atom of chlorine-37 each contain the same number of protons. The atomic weights given inside the back cover of this book are all weighted averages of the isotopes occurring in nature, and these are the figures we shall use henceforth-unless we are specifically discussing one isotope. Chlorine occurs in nature as 75.53% chlorine-35 (34.97 amu) and 24.47% chlorine-37 (36.97 amu), so the weighted average of the isotope weights is The average is weighted according to the percent abundance of the isotopes. (For example, the atomic weight of chlorine-37 is 36.966, which is rounded to 37.) If there are several isotopes of an element in nature, then of course the experimentally observed atomic weight (the natural atomic weight) will be the weighted average of the isotope weights. Since mass losses upon formation of an atom are small, the mass number is usually the same as the atomic weight rounded to the nearest integer. The chlorine isotope has 17 protons and 20 neutrons: Protons:Įach isotope of an element is characterized by an atomic number (total number of protons), a mass number (total number of protons and neutrons), and an atomic weight (mass of atom in atomic mass units). Compare this with the actual atomic weight of this isotope as given in Table 1-2. Helium nucleus or alpha particle, He 2 or αĬalculate the expected atomic weight of the isotope of chlorine that has 20 neutrons. The terms atomic weight and molecular weight are universally used by working scientists, and will be used in this book, even though these are technically masses rather than weights. Nuclei that have too many of either kind of fundamental particle are unstable, and break down radioactively in ways that are discussed in Chapter 23. The more protons, the greater the ratio of neutrons to protons to ensure stability. For a nucleus to be stable, the number of neutrons should (for the first few elements) equal or slightly exceed the number of protons. In discussing these isotopes, we use the. Hence the isotope symbols are usually written without the subscript: 35Cl and 37Cl. Strictly speaking, the subscript is unnecessary, since all atoms of chlorine have 17 protons. The symbols for the two naturally occurring isotopes of chlorine then would be Cl and Cl. To write the symbol for an isotope, place the atomic number as a subscript and the mass number (protons plus neutrons) as a superscript to the left of the atomic symbol. Only two chlorine isotopes exist in significant amounts in nature, those with 18 neutrons (75.53% of all chlorine atoms found in nature), and those with 20 neutrons (24.47%). All atoms of chlorine (Cl) have 17 protons, but there are chlorine isotopes having 15 to 23 neutrons. Four isotopes of helium (He) are shown in Figure 1-1. 
These differing atoms of the same element are called isotopes. \)Īlthough all atoms of an element have the same number of protons, the atoms may differ in the number of neutrons they have (Table 1-2).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
